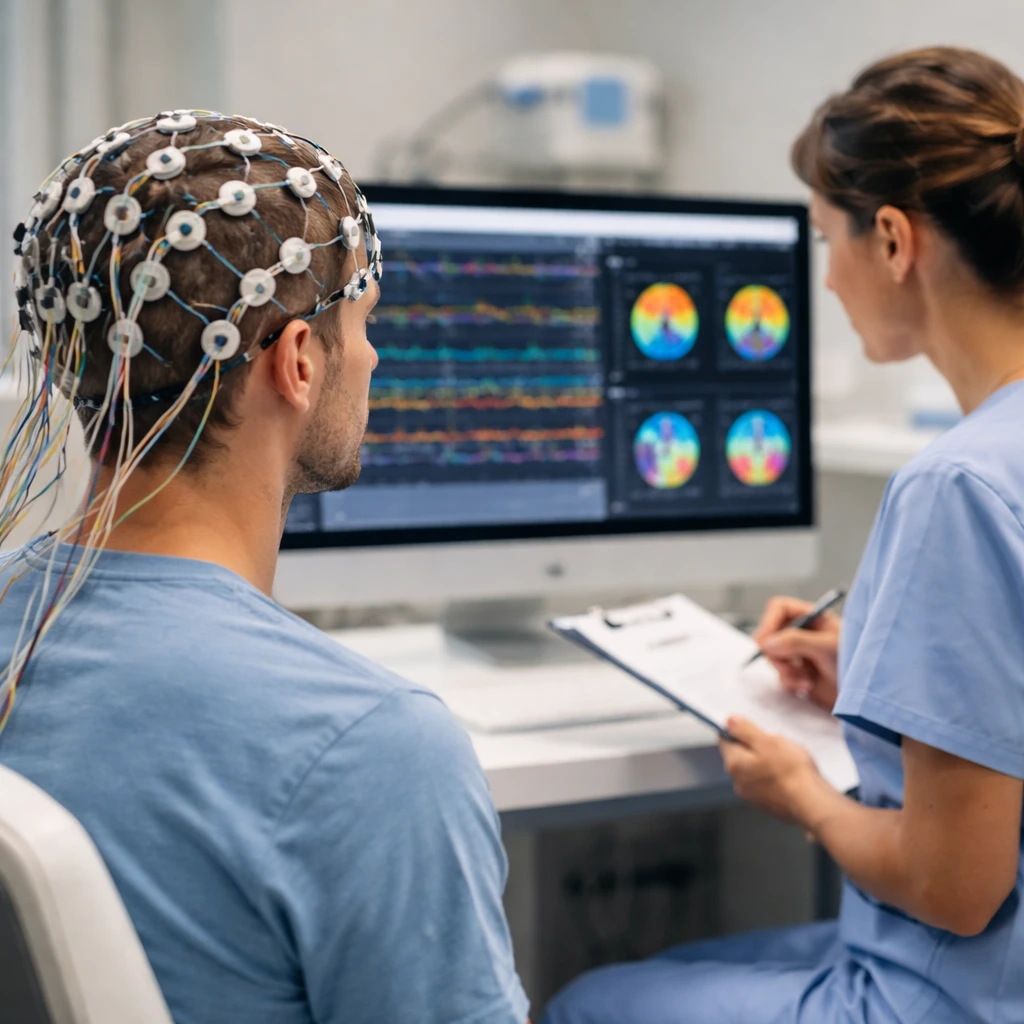
Firefly Neuroscience Inc (NASDAQ:AIFF) saw its share price jump 47.7% on Friday after the company disclosed research progress suggesting its AI-enabled EEG/ERP technology could help distinguish among the three main subtypes of attention-deficit/hyperactivity disorder.
The company said its Evoke System - which is cleared under FDA 510(k) - gathered Resting EEG and Cognitive EEG brain scans that may assist clinicians in identifying whether a patient presents with the hyperactive and impulsive subtype, the inattentive subtype, or the combined subtype of ADHD. Firefly noted that the U.S. treatment market for ADHD is estimated at over $10 billion.
Firefly framed the technology as a complement to current, subjective symptom checklists by offering neural signal-based precision. Company materials say this approach could help determine appropriate treatment types, guide dosage decisions, and provide an objective way to monitor whether interventions are working at the neurological level. The filing underlines that more than 22 million Americans are diagnosed with ADHD, yet an objective biological marker that distinguishes subtypes has not been established.
The firm pointed to peer-reviewed research underpinning its methodology, citing a study published in Frontiers in Psychiatry that demonstrated the diagnostic utility of EEG-based brain activity flow pattern analysis in patients with ADHD. In describing the potential clinical value, Gil Issachar, Firefly's Chief Technology Officer, said:
"Firefly’s EEG/ERP platform can leverage subtype-specific biomarker profiles to inform personalized treatment recommendations, potentially improving outcomes for the millions currently on generalized ADHD treatment plans. We believe that the technology also opens the door to ongoing treatment monitoring - allowing clinicians to objectively track whether an intervention is working at the neurological level."
The announcement also reiterated Firefly's longer-term technical objectives. The company said it is building an EEG/ERP brain foundation model trained on a repository of over 191,000 brain scans, and it has access to NVIDIA GPU acceleration to support next-generation EEG/ERP processing.
Below are concise takeaways, key points and risks drawn directly from the company's disclosure and supporting materials.